| Amazing Men and their Magical Machines |
|
The Gas Laws
There are three equations called the gas laws which govern the relationships between the thermodynamic temperature, the absolute pressure and the volume of gasses.
The first equation is called Boyles Law which describes the inverse relationship between pressure and volume in a closed system if the temperature remains constant.
Boyles Law states that: (P1)(V1)=(P2)(V2)
The second is called Gay-Lussac's Law which describes the relationship between temperature and pressure if the volume is constant.
Gay-Lussac's Law states that: (P1/T1)=(P2/T2)
The third equation is called Charles's Law which describes the relationship between Volume and Temperature if the pressure is constant.
Charle's Law states that: (V1/T1)=(V2/T2)
These three Laws can be used to make the Combined Gas Law which relates all three properties to each other.
Combined Gas Law states that: [(P1V1)/T1]=[(P2V2)/T2]
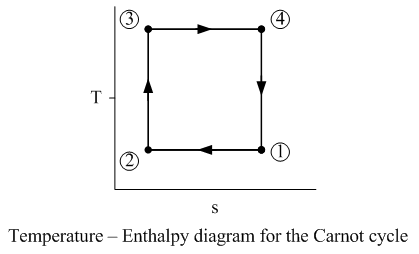
Nicolas Leonard Sadi Carnot came up with the Carnot cycle which is a thermodynamic cycle based on the hypothetical Carnot heat engine. An engine capable of performing like a Carnot engine would be very efficient. The Carnot cycle describes the perfect cycle of the working fluid. This can be shown on a Temperature – Entropy (T-s) diagram as shown below.
However due to material and gas restrictions the chances of the Stirling engine ever reaching this perfection are very low.